 
There are no charges on phosphorus atom and hydrogen atoms. Therefore, then mark that electron pair on center atom phosphorus.Therefore, we cannot mark that electrons pair on hydrogen atoms. But in PH 3, hydrogenĪtom are the outside atoms which cannot keep more than two electron in its last shell. Usually, those remaining electron pairs should be started to mark on outside atoms.There are already three P-H bonds in the above drawn sketch.Remember that, there are total of four electron pairs. Now, we can draw the sketch of PH 3 to describe how atoms are attached with each other.Īfter determining the center atom and sketch of PH 3 molecule, we can start to mark lone pairs on atoms. Therefore, phosphrus atom should be the center atom of Phosphorus, which atom has the highest valence? Maximum valence 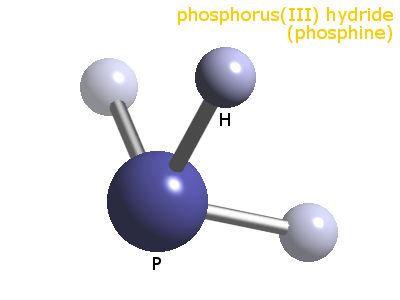
To be the center atom, ability of having greater valance is important. Total electron pairs are determined by dividing the number total valence electrons by two. Pairs = σ bonds + π bonds + lone pairs at valence shells
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
